Application Data Sheet – Stainless Steel
Hydrostatic Testing of Welded Stainless Steel Fabrications
Selection and Treatment of Water and Conduct of the Test, to Maintain the integrity of the System 1.0 Introduction to Hydrostatic TestingWelded fabrications are frequently hydrotested for pressure tightness. Unless properly conducted, this of pressure testing can induce problems. It is important to avoid initiating corrosion of the fabrication by subjecting it to critical conditions outside the initial design constraints. Where the vessel is to handle materials which are later ingested, contamination must be avoided. Damage may be caused by the induction of latent corrosion problems during and after a hydrostatic test. The damage may not become apparent until some weeks or months later, and may very difficult and costly to repair. However, if simple precautions are taken, a hydrostatic test can be conducted without risk to the equipment. The precautions mainly relate to the quality and the treatment of the water used, and how the equipment is treated after hydrotesting. And hydrotesting even offers an opportunity to improve the quality of the fabrication under test pressure. This note gives general advice on how to conduct a hydrostatic test without damaging the equipment. However, the subject is complex, and the onus remains with the tester to ensure that the practices used will not cause problems. There are no published standards or codes of conduct reporting industry consensus on hydrotesting stainless steels, although several give general advice (see boxes below and on page 8). Please consult Austral Wright Metals for further information. 2.0 Corrosion of Stainless Steels after Hydrostatic TestCorrosion failures of fabricated stainless steel systems soon after hydrotesting may result from one or more of three causes:
Pitting and crevice corrosion can result from attack by the water used for hydrostatic pressure testing. The equipment is designed for particular conditions, and the grade and surface finish are selected from knowledge of the proposed temperature, solute concentrations, pH etc. If the water conditions used for testing are more aggressive, corrosion may occur. Unfortunately, corrosion by these mechanisms is localised, and proceeds in isolation from the bulk solution conditions: once established, it can proceed in conditions which are otherwise not aggressive. Failure may occur by local penetration 3 – 12 months after initiation of the corrosion.
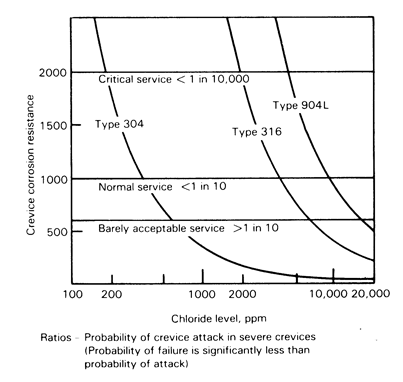
Figure 1: Probability of crevice corrosion attack as a function of chloride content of water. The line for the super austenitic 904L also represents the duplex grade S31803 (2205) [1].
|
Time and temperature are also important in the development of pitting & crevice corrosion, and water with higher chloride content may be used safely if the contact time and temperature are limited.
Apart from the initial quality of the water, its aggressiveness can be increased by increasing temperature, by oxygen starvation under particles deposited on the surface of the stainless, and by concentration of chlorides in pools of water left in the system after testing. See section 5 for precautions to be taken after hydrostatic pressure testing.
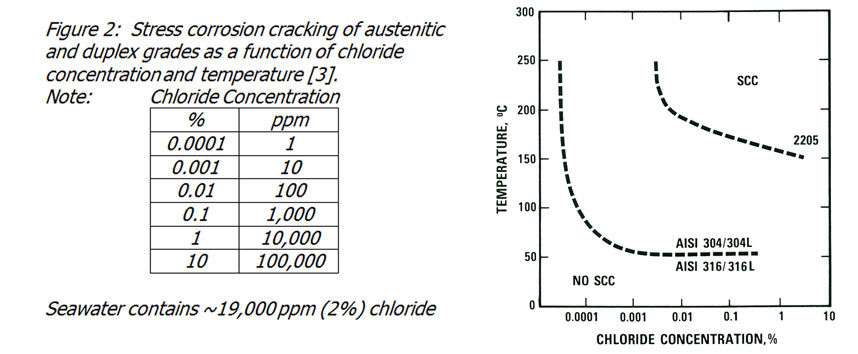
Stress corrosion cracking is a failure mechanism which can cause failure very rapidly. Stainless steels are not especially susceptible to stress corrosion failure in waters, but it may be encountered at high chloride levels and temperatures in austenitic grades such as 304 and 316. The duplex grades (e.g. 2205) are effectively immune from this form of failure in waters.
The austenitic grades are also effectively immune to stress corrosion cracking in waters at temperatures below about 50°C (figure 2). Since hydrotesting is usually performed at ambient temperature, the problem is unlikely to arise. However, equipment left in strong sunlight for a long period could get to this temperature, particularly in exposed pipe runs. Care should be taken to avoid excessive temperatures.
Microbiological Corrosion (MIC) is a mechanism which is increasingly being recognised as contributing to corrosion of metals [4]. Penetration rates as high as 3.2 mm in a month have been reported for 304L, and in four months for 316L [5]. Micro-organisms, primarily bacteria, can alter the micro-environment to which the steel surface is exposed, by concentrating aggressive species, and by restricting the access of oxygen to the surface.
Sulphate reducing bacteria (anaerobic) metabolise sulphate ions to sulphide ions, while sulphate oxidising bacteria (anaerobic) can metabolise sulphur compounds to sulphuric acid. Both are highly aggressive to stainless steel.
The most common MIC problems with stainless steels are caused by iron utilising bacteria, which concentrate chloride as iron and manganese chloride, to much higher concentrations than the level in the bulk of the water. This can easily far exceed the chloride levels mentioned above, and the resulting corrosion mechanism is often pitting and/or crevice corrosion: crevices, especially at gaskets, are one favoured site for this form of corrosion. Another common site is at the weld. Numerous studies have shown that properly treated welds are not generally preferential sites for corrosion, but it is probable that surface imperfections such as oxide or slag inclusions, porosity, ripples, et cetera may be easy sites for bacteria to colonise.
MIC almost invariably occurs with raw (untreated) fresh water sources such as reservoirs, rivers, bores or wells, or fresh waters inadequately treated to prevent MIC.
Stagnant and low flow conditions promote attachment of bacteria to the metal surface. This is followed by colonisation and formation of discrete deposits, starting as a biofilm, and frequently developing into a bulky deposit. The composition of the deposit is significantly different from and more corrosive than the bulk water. Localised attack manifests as [4]:
- large subsurface cavities or tunnels, frequently at or adjacent to welds
- as broad, open pits in crevices such as gasketed flanged joints
- as broad conical pits with terraced sides
MIC can be prevented by draining and drying equipment after hydrotesting. The bacteria require time to grow, and no significant damage should be caused during the restricted time of hydrotesting.
This supposes that drying is complete and thorough, with no possibility of pools being left in the system. It is more conservative to also treat the water to disinfect it of bacteria before use. This is discussed in more detail in section 4.
3.0 Water Quality for Hydrostatic Pressure Testing
NHMRC/ARMCANZ Australian Drinking Water Guidelines (ADWG) give a guideline maximum value for chloride of 250 ppm, and most potable water supplies in Australia conform to this limit. However, some supplies do not, especially where there is a significant proportion of bore water in the supply, and at times of drought, when the Water Authority may use alternative supplies. The Water Authority has information on the chloride level of the water supplied.
Since the 200 (304), 2,000 (316) and 4,000ppm (2205) chloride levels discussed above as being acceptable for avoiding pitting & crevice corrosion (figure 1) are for extended service, use of water somewhat above these levels should not cause problems at the shorter times used in hydrostatic pressure testing. Moving from the ‘critical service’ line to the ‘normal service’ line in figure 1 gives limiting chloride content of 400 ppm for 304, 4,000 ppm for 316, and 8,000 ppm for 2205. Obviously, this requires the testing time to be kept short, and the equipment to be drained and dried after testing – see next section.
Other properties of potable water, such as pH, dissolved oxygen and hardness, are unlikely to cause corrosion problems for stainless steel.
4.0 Water Treatment
Where water is to be obtained from a source other than a reticulated potable supply, the chloride levels given above should still be adhered to, as a minimum requirement. Water analysis labs will advise on how to take samples, and will analyse the samples for a hundred dollars or so. They may be found under “Analysts” in the local Yellow Pages Telephone Directory.
Where it is essential to use water sources with chloride contents higher than these limits, and also for non potable supplies, the pH should be adjusted into the alkaline range, say 7.5 – 10. Caustic soda (NaOH) can be used for this, and has a double benefit in that the hydroxide ion inhibits corrosion of stainless. Sulphate is also a useful inhibitor, and may be added to low sulphate, high chloride waters, up to about 100 ppm.
Austral Wright Metals are able to predict the corrosivity of waters, using software developed by the Nickel Development Institute. Contact Austral Wright Metals for a copy of the software, or predictions of corrosivity are available to customers, requiring the input data as shown in Appendix 1 to produce the outputs shown in the Appendix. |
Potable water supplies should be free of particulates, but other sources should be screened or settled and filtered. A filter passing particles up to 25 micrometres is probably adequate.
Sanitisation or disinfection of the water is essential to remove the risk of MIC following a hydrostatic pressure test. It can be carried out by a variety of methods, some of which also carry risks of making the water more aggressive for other forms of corrosion. Potable water from properly conditioned town supplies do not require further sanitisation.
The most common sanitisation method is chlorination, and the most common method of introducing chlorine is with a hypochlorite salt, which is available as a dry powder and as liquid solution, most conveniently from swimming pool supply shops. The addition required is usually small, and online additions are difficult to control. It is best, where possible, to disinfect the water as a batch before introducing it to the system under a pressure test, adding the disinfectant last and ensuring good mixing. As a guide, Table 1 [6] gives the quantity of sodium hypochlorite required for specific volumes of testing water. More polluted water supplies may need more hypochlorite. The chlorine residual achieved can be checked with a normal colorimetric swimming pool chlorine test kit.
Free chlorine levels at entry to the system should be limited to 2ppm for grade 304, 5ppm for grade 316 [7]. The 2 ppm limit for residual free chlorine is also applicable to any copper alloys in the system.
Short term levels of free chlorine of up to 25 ppm can be tolerated by 316, provided there is thorough post cleaning treatment rinse, using water with the residual chlorine level maxima already mentioned [7].
After hydrotesting it is prudent to measure the chlorine residual of the last water to be drained from the system. If this is below 0.2ppm, or shows evidence of sludge, the system should be flushed with properly treated sanitised water without delay.
Other disinfectants which may be used include chlorine gas, chlorine dioxide, ozone and ultraviolet light. The former are subject to the same restrictions on free chlorine residuals, while ozone and ultraviolet light pose no risks for stainless steel. Indeed, ozone may be beneficial for corrosion resistance.
Table 1: Quantities of sodium hypochlorite required for residual disinfectant in flushing & test water [6].
| Volume of Test Water | Volume of 12.5% Sodium Hypochlorite to produce 2 ppm residual free Chlorine in Clean Water |
| Litres | Millilitres |
| 10 | 0.2 |
| 50 | 1.0 |
| 100 | 2.0 |
| 1,000 | 20 |
| 10,000 | 200 |
| 50,000 | 1000 |
5.0 Precautions after Hydrotesting – Drying
Equipment left wet after testing is at risk because the chloride levels of pools of water will rise as the water evaporates: PCC may initiate at these high chloride levels.
To prevent chloride concentration after a hydrostatic pressure test, the equipment should be thoroughly dried and sealed if it is to be left for more than a day or two before being put into service. This can be achieved by draining the water out (properly designed equipment will allow for complete drainage), then blowing dry air through the wet spaces. When the humidity of the air is high, this process should be completed when the work is at its coolest (i.e. at night), to prevent condensation when the work cools.
Drying the equipment also stifles any microbiological infections, which will not progress on a dry surface.
6.0 Possible Beneficial Effects of Hydrostatic Pressure Testing
ASTM A967 “Chemical passivation treatments for stainless steel parts” [8] gives a series of treatments for improving the corrosion resistance of stainless steel surfaces by passivation: ‘……. defined as the chemical treatment of a stainless steel with a mild oxidant, such as a nitric acid solution, for the purpose of removal of free iron or other foreign matter, but which is generally not effective in removal of heat tint or oxide scale on stainless steel.’
By mixing a suitable oxidant with the hydrotesting water to passivate the surface, it should be possible to improve the overall corrosion resistance of the system, especially where a passivation treatment has not previously been applied to the surfaces. These passivation treatments require that the surfaces be pretreated to be substantially free of oil, grease, rust, scale and other foreign matter.
The standard specifies five alternative treatments with nitric acid, but the most practical in this context is 20 to 45% nitric acid at 21 to 32°C for a minimum of 30 minutes, followed by a clean water rinse.
Probably more practical for hydrotesting is one of the five citric acid based solutions, using 4 to 10% citric acid for a minimum of 20 minutes at 21 to 50°C.
Care should be taken to evaluate the likely effect on the entire system: there may be carbon steel or copper alloys which would be unduly attacked by the acid.
After testing, the system should be thoroughly rinsed, with water of adequate quality as detailed above. The acid effluent must be neutralised before disposal. It is still good practice to dry the system within a day or two after testing.
While these treatments offer potential improvement to the corrosion resistance of the steel surfaces contacted, they are by no means essential, and the benefits may be outweighed by the costs of the chemicals, effluent neutralisation and rinsing.
7.0 Treatment of the Stainless Surface before Hydrotesting
In all the discussion in this note it has been assumed that all welds in contact with the hydrotesting water have been made or treated to minimise or eliminate the visible welding oxide and adjacent chromium depleted zone from the surface. This is equivalent to external surfaces on the weld seams of Quality B, Surface Finish II, as defined in AS/NZS 1554.6:1994, “Welding stainless steels for structural purposes” [9]. The Grade II finish may be achieved by (a) acid pickling, (b) stainless steel wire brushing, (c) buffing or linishing to a specified abrasive grit size, or (e) inert gas purging.
Where option (c), linishing, is specified, best corrosion performance is obtained with a surface roughness (Ra) of 0.5 micrometres or smoother. This can usually be achieved with a worn belt of grit size 180#, although 220# is more certain.
Option (d) provided in AS/NZS 1554.6 of abrasive grit blasting is not recommended.
Where option (e) inert gas purging is used, AWS D18.1 “Specification for welding of austenitic stainless steel tube and pipe systems in sanitary (hygienic) applications” [10] provides guidance and colour standards on the level of oxidation of the surface which is tolerable.
ASTM A380 “Standard practice for cleaning, descaling and passivation of stainless steel parts, equipment and systems” [11] provides detailed guidance on the conditioning of stainless steel surfaces for best corrosion resistance.
There is a reference to hydrotesting in the ASME Power Piping Code B31.1, relative to stainless steel. It appears in Appendix IV under Nonmandatory Corrosion Control for ASME B31.1 Power Piping Systems”, section IV-3.4, precautions for hydrotesting. |
SUMMARY
slope horizontal lines and components to allow self draining, and provide adequate drainage at high and low points of the system
|
References
[1] Nickel stainless steels for marine environments, natural waters and brines, Nickel Development Institute No 11 003, 1993.
[2] RP0182-95 Standard recommended practice for initial conditioning of cooling water equipment, NACE, 1995.
[3] A.J. Sedriks, Corrosion of stainless steels, 2nd Edn, Wiley, 1996.
[4] G. Kobrin et al, Microbiologically influenced corrosion of stainless steels by water used for cooling and hydrostatic testing, Nickel Development Institute No 10 085, 1998.
[5] G. Kobrin, Material Performance, vol 15, No 7, p40, July 1976.
[6] DR 02014, Copper pipe and fittings – installation and commissioning, Standards Australia 2002.
[7] Stainless Steel Advisory Service Information Sheet No.4.92, Issue 02, March 2001, British Stainless Steel Association, UK.
[8] ASTM A967 Chemical passivation treatments for stainless steel parts.
[9] AS/NZS 1554.6:1994 Welding stainless steels for structural purposes, Standards Association of Australia, 1994.
[10] American Welding Society Standard AWS D18.1 Specification for welding of austenitic stainless steel tube and pipe systems in sanitary (hygienic) applications.
Appendix 1
Nickel Development Institute Crevice Corrosion Model
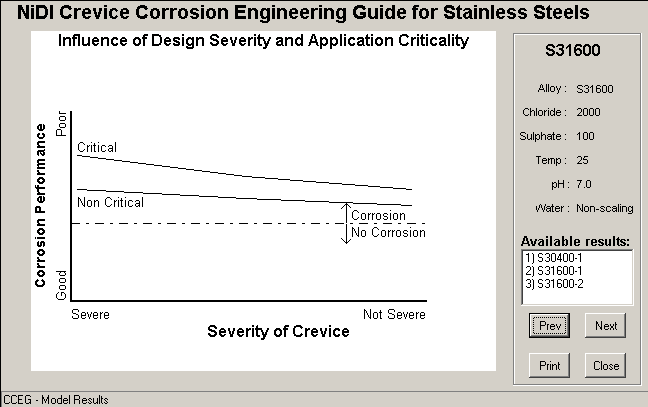
a) Results screen
CCEG MODE 1 SUMMARY
The user has asked the CCEG for guidance on the corrosion of S31600 under the conditions specified below.
Under these conditions the CCEG predicts that corrosion will initiate in DAYS.
| SERVICE APPLICATION | ||
Design Severity: Severe | Application Criticality: Critical | |
| WATER PARAMETERS | ||
| Chloride (ppm): 2000 | Sulphate (ppm): 100 | |
| Hardness (as CaCO3 mg/L): 750 | Alkalinity (as CaCO3 mg/L): 100 | |
| TDS (mg/L): 3448 | pH: 7 | |
| Temp (C): 25 | Scaling pH: 7.00 | Water is Non-scaling |
| Oxygen level (ppm): 7 | ||
| MATERIAL | ||
Form: Default (Wrought) | Fabrication: Welded | Condition: Pickled |
b) Output Screen 1
c) Output Screen 2
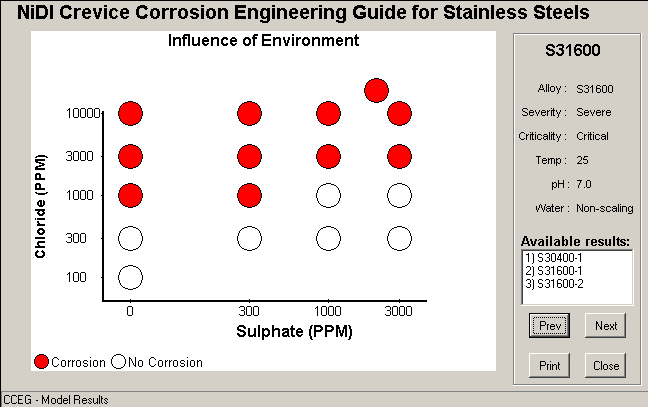
Data for Crevice Corrosion Predictor

| The technical advice and recommendations made in this Product Data Sheet should not be relied or acted upon without conducting your own further investigations, including corrosion exposure tests where needed. Please consult current editions of standards for design properties. Austral Wright Metals assumes no liability in connection with the information in this Product Data Sheet. Austral Wright Metals supplies a comprehensive range of stainless steels, copper alloys, nickel alloys and other high performance metals for challenging service conditions. Our engineers and metallurgists will be pleased to provide further data and applications advice. |